Properties of Ammonia
Properties of Ammonia
Ammonia is toxic. It is a pungent gas that affects the respiratory organs and mucous membranes.
Ammonia is flammable. The ignition limit is 630°C, in the absence of catalytic steel even 850°C.
Ammonia cannot be ignited with a burning shaving, but it can be ignited with a welding torch.
Ammonia is explosive. The lower explosion limit in connection with air is 15.0%, the upper explosion limit is 30.2% (related to 20°C and 1.013 bar).
Why has such a dangerous substance been used as a refrigerant for 120 years?
- The manufacturing costs are low.
- The thermodynamic properties are favorable compared to other refrigerants.
- The ecological values are fine: ODP = 0, GWP = 0.
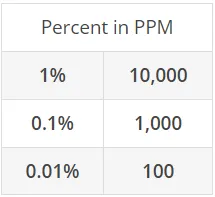
- Perceptions at 5 -10 ppm (volume in air)
ppm - parts per million - the millionth part of a unit
Usable Materials
The main material used is steel and its alloys. Galvanic surfaces are destroyed by ammonia.
Zinc is completely dissolved by ammonia in the presence of water.
Copper cannot be used as a material. Ammonium hydroxide forms in the presence of water. This readily dissolves copper to form the characteristic blue-colored copper-ammonium complex.
Phosphor bronze is the most resistant to ammonia. It is used in shaft seals. Alternative material is carbon and silicon carbide.
Mercury forms explosive mixtures with ammonia, and the addition of bismuth amalgam can help.
What are the concentration limits?
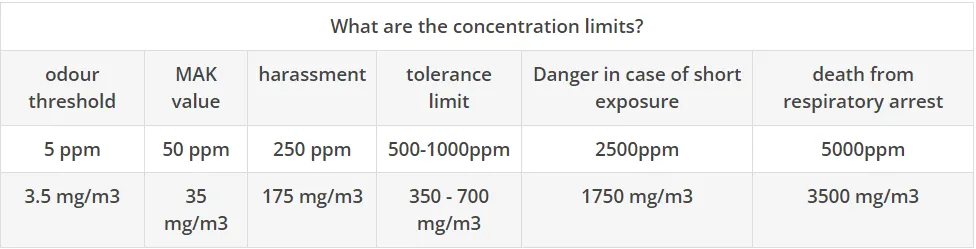
- MAK value 50 ppm
- 300 ppm hardly tolerable. Still harmless if exposed for more than an hour.
- 700 - 1,000 ppm unbearable. Prolonged exposure will damage the respiratory tract.
- 2,000 - 3,000 ppm fatal after 0.5 - 1 hour. Corneal inflammation occurs in the eyes.
- 5,000 - 6,000 ppm leads to blindness and death after 30 minutes








